The Laboratory of Nanostructures at IWC PAS has developed an innovative technology for the synthesis of hydroxyapatite* nanoparticles - a synthetic equivalent of the mineral responsible for bone formation. Thanks to this technology, it is possible to obtain nanoparticles of precisely controlled size.
* Patent-protected technology: PAT.235292 „Method for of producing nano-plates of synthetic hydroxyapatite and nanopowder comprising a synthetic hydroxyapatite nano-plates”

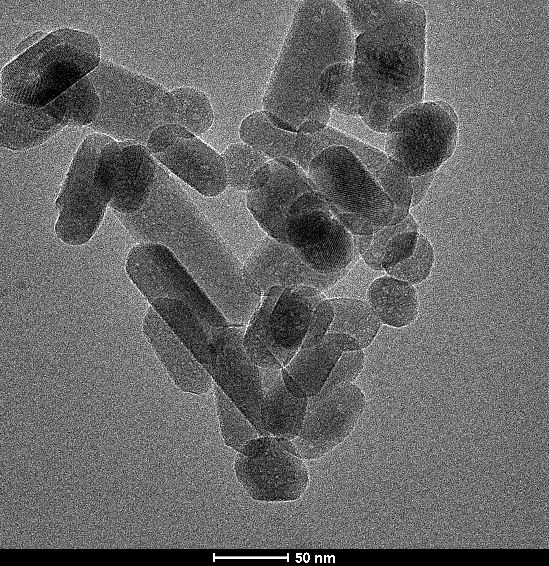
Product images from Transmission Electron Microscopy
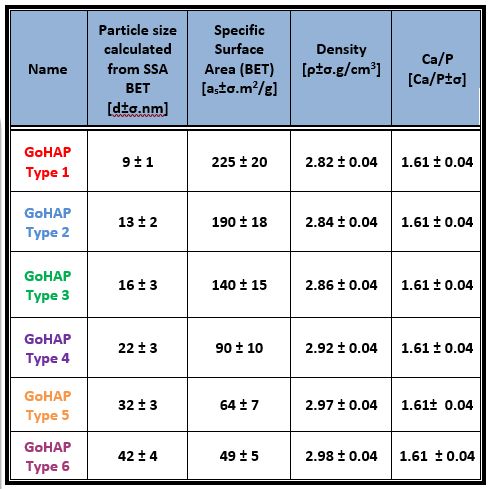 The laboratory produces 6 types of GoHAPTM hydroxyapatite with different nanoparticle sizes. Hydroxyapatite is obtained under clean conditions using pharmacopeial purity substrates and in Class I water environment. This material can be used as a filler for bone defects to stimulate their faster regeneration.
The laboratory produces 6 types of GoHAPTM hydroxyapatite with different nanoparticle sizes. Hydroxyapatite is obtained under clean conditions using pharmacopeial purity substrates and in Class I water environment. This material can be used as a filler for bone defects to stimulate their faster regeneration.
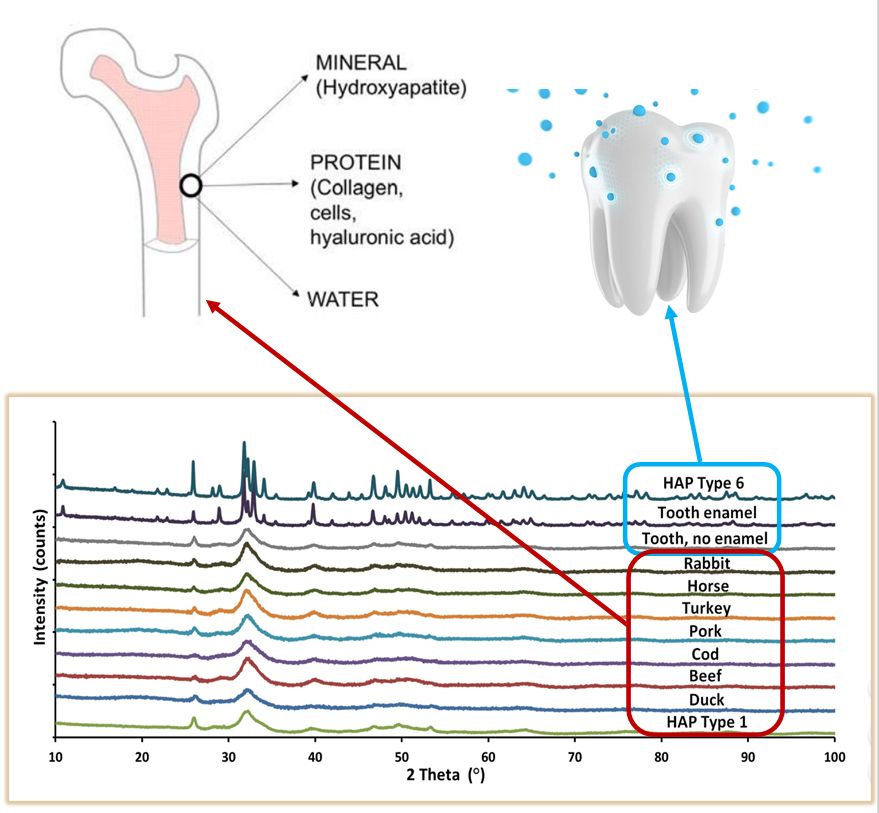
Figure 1 Phase analysis of hydroxyapatite by X-ray diffraction
- SYNTHETIC BONE MINERAL FOR ENHACEMENT BONE TISSUE REGENERATION.
- THE GoHAP NANOMATERIAL PERFECTLY CORRESPONDS TO NATURAL HYDROXYAPATITE, WHICH IS A COMPONENT OF BONE.
- A NANOMATERIAL IN POWDER FORM WHICH, WHEN COMBINED WITH BLOOD, FORMS A PLASTIC PASTE .
- POSSIBLE APPLICATIONS IN ORTHOPEDICS AND DENTISTRY.
- IT MAY BE USED FOR THE TREATMENT OF COMPLEX BONE FRACTURES OR OTHER SURGICAL TREATMENT OF BONE TRAUMA.
- INNOVATIVE PRODUCT IN THE CERTIFICATION AND IMPLEMENTATION PHASE FOR MEDICAL DEVICES ACCORDING TO EUROPEAN REQUIREMENTS.
PRODUCT DESCRIPTION:
The advantage of GoHAP nanopowder hydroxyapatite over other apatites is due to its size and crystal structure. The precise control of particle size at the synthesis stage is an innovation on a global scale. Through fully controlled microwave synthesis it is possible to produce hydroxyapatite nanopowder with almost identical morphology in comparison to the hydroxyapatite naturally contained in bone. Currently, most bone substitute materials available in the medical device market are micrometric sized, and in the case of hydroxyapatite nanomaterials, materials with nanoparticle sizes above 40 nm are the most common.
The particle size of biomaterials, and nanomaterials in particular, has a huge impact on their properties - both in terms of the rate of degradation in the body and the activity of the nanoparticles in accelerated bone repair. Natural hydroxyapatite, which is a component of bone, has the form of nanoparticles with a needle-like shape and stoichiometric calcium deficiency. The hydroxyapatite produced in the Laboratory of Nanostructures IHPP PAS is an almost perfect match to natural hydroxyapatite in both particle size and shape. The high similarity was also demonstrated in X-ray analysis (Figure 1). GoHAP, due to its much smaller grain size, is more effective in bone-forming activity compared to micrometric apatites.
In preliminary studies, the effect of grain size on the solubility of GoHAP in an environment simulating conditions in the human body was demonstrated. The solubility of hydroxyapatite increases with decreasing grain size. The higher the solubility of hydroxyapatite, the faster the release of calcium ions into the body, which provides a signal for the formation and proliferation of bone-forming cells. Cytotoxicity tests performed according to ISO 10993-5 (Biological evaluation of medical devices - Part 5: In vitro cytotoxicity tests) confirmed the absence of cytotoxic effects on cells. In addition, this material was used for treatment of dogs in a reference center.
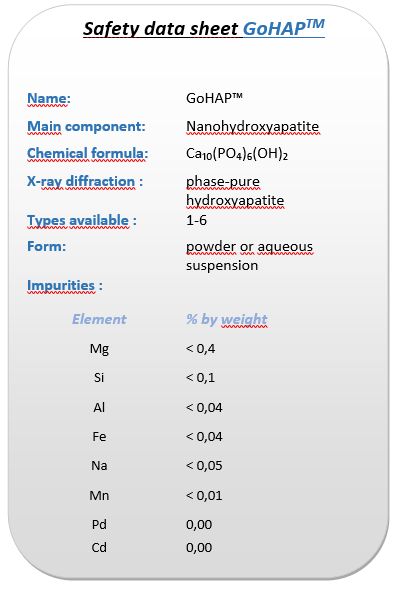
Selected cases of different applications of GoHAP material and their treatment effects are presented below. Treatments consisted of the application of GoHAP nanopowder in dry form or mixed with blood/bone marrow taken from the patient to the bone defect. This type of treatment was used in the following cases: rheosteosynthesis (fusion of bone fragments after car accidents), knee joint stabilization (Figure 2) and arthrodesis (Figure 3). In each of these cases, the use of GoHAP nanomaterial had a significant effect on accelerating the bone defect reconstruction and therefore on accelerating the healing process of the dogs.
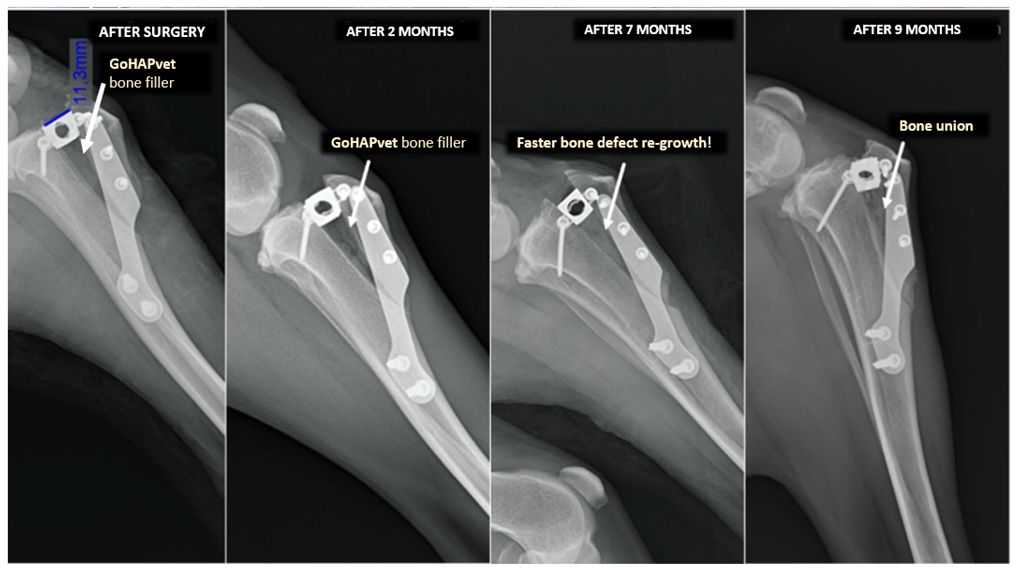
Figure 2 X-ray images of knee joint stabilization after ligament rupture. The treatment included a titanium plate and stabilizing cage and filling the gap with GoHAP nanohydroxyapatite; The image shows the phases of overgrowth of the defect after application of the GoHAP nanohydroxyapatite material.
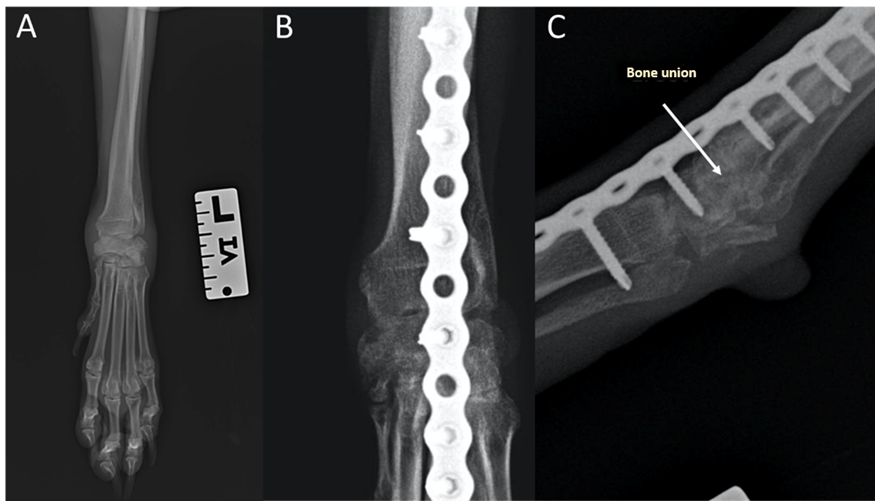
Figure 3 X-ray images: A) paw after ligament rupture; B) stabilized wrist bones; C) adhesion several months after union with GoHAP™ nanopowder.
GoHAP COATINGS OSTEOCONDUCTIVE HYDROXYAPATITE LAYERS ON IMPLANTS
In addition, the Laboratory of Nanostructures IHPP PAS has developed and patented technology for depositing layers of hydroxyapatite nanoparticles on implant surfaces. It is possible to apply GoHAP layers to any surface - from smooth surfaces to porous structures. Also the type of the implant material is not a limitation for the process of applying hydroxyapatite layers - thanks to an innovative method of sonochemical deposition of layers we can apply coatings on materials such as titanium, ceramics, glass, polymers, as well as temperature-sensitive biodegradable polymers.
The osteoinductive properties of GoHAP nanoparticle layers on implants were confirmed in in vivo studies. In the in vivo studies, GoHAP nanoparticles were applied as a layer on a bone scaffold. The results confirmed that the application of GoHAP nanoparticles layer on the polymer implant highly accelerated the bone defect overgrowth process. Figure 4 shows the example of coating deposited with GoHAP hydroxyapatite nanoparticles. Figure 4 (B, C) shows the histopathology results. Bone defect overgrowth when a polymer scaffold implant is used (Figure 4 B) and a polymer scaffold coated with GoHAP nanoparticles (Figure 4 C). The new tissue was designated by the letters NB.
Figure 4 (A) Example of coating deposited with GoHAP hydroxyapatite nanoparticles; (B,C) Histopathology of a cross section of a bone scaffold: (B) made of PCL; (C) made of PCL with GoHAP nanohydroxyapatite coating.
If you are interested please contact us!
 Laboratory of Nanostructures Institute of High Pressure Physics PAS
Laboratory of Nanostructures Institute of High Pressure Physics PAS